
The Bath Ankylosing Spondylitis Disease Activity Index (BASDAI) is a validated, self-administered 6-item questionnaire that measures disease activity in ankylosing spondylitis (AS) and axial spondyloarthritis (axSpA). Developed by Garrett et al. in 1994 at the Royal National Hospital for Rheumatic Diseases in Bath, England, the BASDAI assesses five symptom domains — fatigue, spinal pain, peripheral joint pain/swelling, localized tenderness (enthesitis), and morning stiffness — using 0–10 visual analog scales, producing a composite score from 0 to 10, where ≥4 indicates active disease requiring treatment escalation (Garrett et al., Journal of Rheumatology, 1994). The BASDAI demonstrates excellent test-retest reliability (r = 0.93) and sensitivity to change, making it the most widely used disease activity measure in AS clinical practice and research. Zentake's digital BASDAI enables HIPAA-compliant administration with automated scoring and longitudinal tracking.
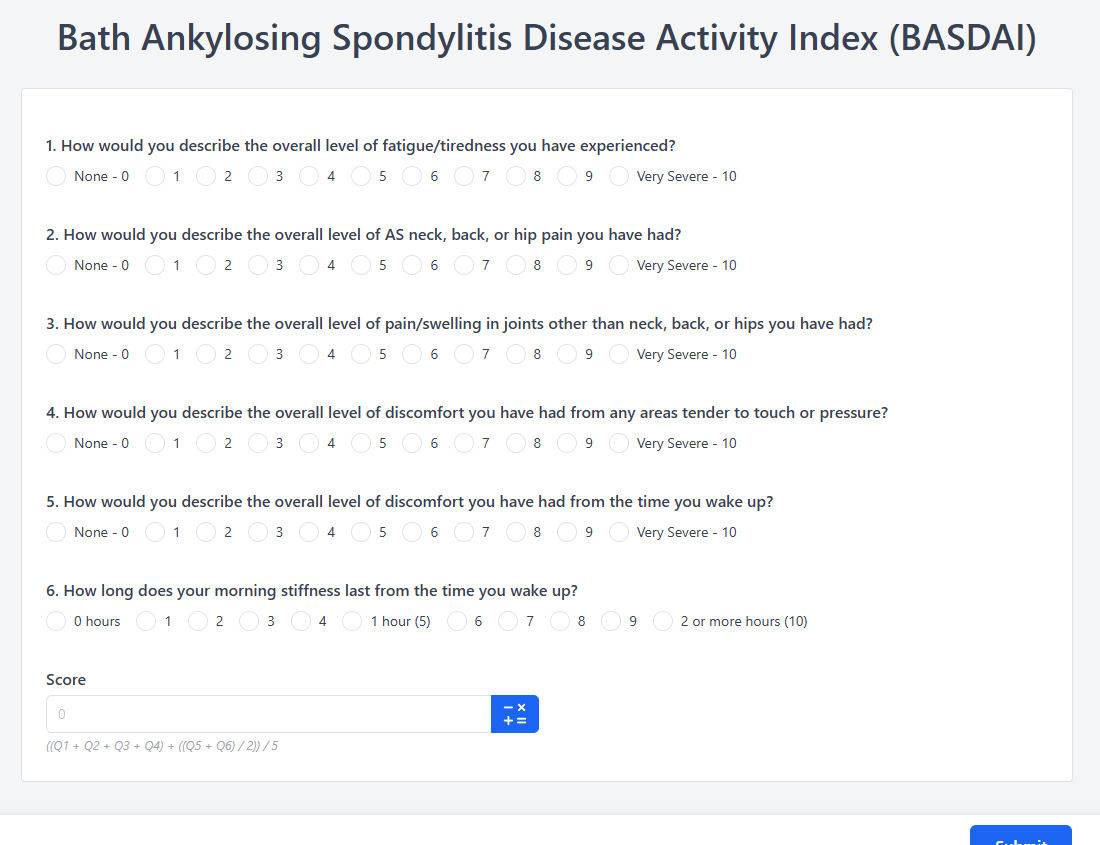
The BASDAI consists of six questions, each rated on a 0–10 visual analog scale (VAS) or numerical rating scale (NRS). The six items assess: (1) fatigue/tiredness, (2) AS neck/back/hip pain, (3) pain/swelling in joints other than neck/back/hips, (4) discomfort from areas tender to touch or pressure, (5) severity of morning stiffness, and (6) duration of morning stiffness (0 = 0 hours, 10 = 2+ hours).
To calculate the total BASDAI score, first average the scores of questions 5 and 6 (the two morning stiffness items). Then add this average to the scores of questions 1 through 4. Finally, divide the total by 5 to produce a final score ranging from 0 to 10.
Zentake automatically calculates the BASDAI composite score upon form submission, eliminating manual computation errors and providing instant results to clinicians.
Step 1: Select the assessment. Choose the BASDAI from your Zentake template library and assign it to the patient. The form can be sent via email link, SMS, or completed on an in-clinic tablet before the appointment.
Step 2: Patient self-completion. The patient independently rates each of the six items on a 0–10 scale based on their symptom experience over the past week. The BASDAI takes an average of 67 seconds to complete (range: 30–120 seconds).
Step 3: Review auto-scored results. Zentake automatically calculates the composite BASDAI score and presents it alongside individual item scores. Clinicians can review results before the patient enters the consultation room.
Step 4: Compare longitudinal trends. Use Zentake's measures tracking to compare the current BASDAI score against previous assessments, identifying trends in disease activity over weeks or months of treatment.
Step 5: Document and integrate. Export results directly to your EMR system or generate a PDF summary for the patient's chart. All data is stored securely in Zentake's HIPAA-compliant cloud.
Scoring: Paper BASDAI requires manual averaging of morning stiffness items and calculation of the composite score, which is error-prone. Digital BASDAI through Zentake calculates scores instantly and accurately upon submission.
Completion: Paper forms must be distributed, collected, and filed manually. Digital forms are sent automatically before appointments and completed on any device in about 67 seconds.
Delivery: Paper forms are only available in-clinic. Zentake's digital BASDAI can be completed at home, in the waiting room, or on a mobile device, improving pre-visit preparation.
Data Storage: Paper forms require physical filing and are vulnerable to loss or damage. Zentake stores all data in encrypted, HIPAA-compliant cloud storage with full audit trails.
Longitudinal Tracking: Comparing paper BASDAI scores over time requires manual chart review. Zentake automatically graphs score trends, making disease activity patterns immediately visible.
Security: Paper forms can be accessed by unauthorized individuals. Zentake employs role-based access controls, encryption, and audit logging to protect patient data.
Integration: Paper results must be manually transcribed into EHR systems. Zentake integrates directly with leading EMR platforms for seamless data transfer.
Cost: Paper-based workflows involve printing, storage, and staff time for manual processing. Zentake's digital solution reduces these costs while improving accuracy and efficiency.
What does the BASDAI measure?
The BASDAI measures disease activity in ankylosing spondylitis and axial spondyloarthritis across five symptom domains: fatigue, spinal pain, peripheral joint pain and swelling, enthesitis (localized tenderness), and morning stiffness (both severity and duration). It produces a composite score from 0 to 10, where higher scores indicate greater disease activity. Zentake's digital version captures all six items and calculates the composite score automatically.
How do you score the BASDAI?
Average the scores of questions 5 and 6 (morning stiffness severity and duration), then add the scores of questions 1 through 4 (fatigue, spinal pain, peripheral joint pain, enthesitis). Divide the total by 5 to produce the final BASDAI score (0–10). A score of 4 or above indicates active disease. Zentake performs this calculation instantly upon form submission.
How long does the BASDAI take to complete?
The BASDAI takes an average of 67 seconds to complete, with most patients finishing within 30 to 120 seconds. It consists of only 6 questions on visual analog or numerical rating scales. Zentake's mobile-friendly digital format enables quick completion on any device before or during clinic visits.
Is the BASDAI free to use?
Yes, the BASDAI questionnaire itself is freely available for clinical use without licensing fees. Zentake provides a ready-to-use digital BASDAI template that includes automated scoring, HIPAA-compliant storage, and EMR integration as part of its platform. Start your free trial at zentake.com.
What population is the BASDAI designed for?
The BASDAI was developed for adult patients diagnosed with ankylosing spondylitis or axial spondyloarthritis. It has been validated across diverse populations internationally and is recommended by ASAS (Assessment of SpondyloArthritis international Society) and EULAR as a core outcome measure in AS clinical care and research.
Who should administer the BASDAI?
The BASDAI is a self-administered questionnaire — patients complete it independently without clinician assistance. Any healthcare professional involved in rheumatology or musculoskeletal care can incorporate it into their workflow, including rheumatologists, primary care physicians, physical therapists, and nurse practitioners. Zentake makes distribution and collection fully automated.
What is the difference between the BASDAI and ASDAS?
The BASDAI relies entirely on patient-reported symptoms, while the Ankylosing Spondylitis Disease Activity Score (ASDAS) combines patient-reported outcomes with laboratory inflammatory markers (CRP or ESR). The ASDAS is increasingly recommended as the preferred composite measure, but the BASDAI remains the most widely used tool in daily clinical practice due to its simplicity and the fact that it does not require blood work.
1. Garrett S, Jenkinson T, Kennedy LG, Whitelock H, Gaisford P, Calin A. A new approach to defining disease status in ankylosing spondylitis: the Bath Ankylosing Spondylitis Disease Activity Index. Journal of Rheumatology. 1994;21(12):2286-2291.
2. Braun J, van den Berg R, Baraliakos X, et al. 2010 update of the ASAS/EULAR recommendations for the management of ankylosing spondylitis. Annals of the Rheumatic Diseases. 2011;70(6):896-904.
3. Machado P, Landewé R, Lie E, et al. Ankylosing Spondylitis Disease Activity Score (ASDAS): defining cut-off values for disease activity states and improvement scores. Annals of the Rheumatic Diseases. 2011;70(1):47-53.
Last updated: March 2026